Why Choose Us?
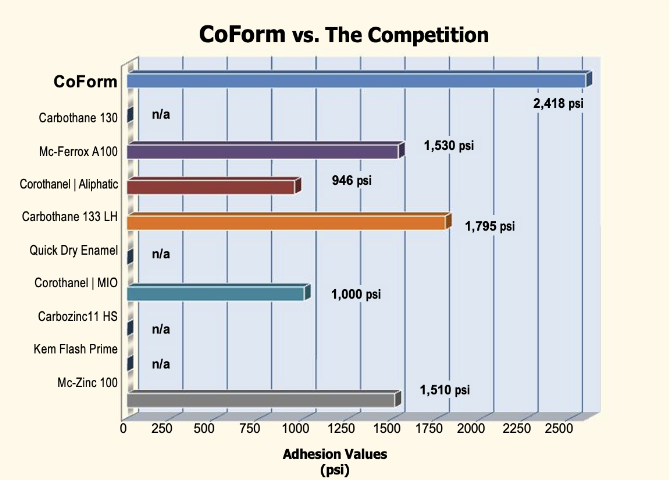
CoForm™ vs. the Competition
PCT’s’s chemists are keenly aware of the physical and chemical properties of CoForm’s perceived competitors such as urethanes, epoxies, acrylics, polyureas, polyvinyls, alkyds, organic zinc-rich coatings, and now, waterborne formulations. For example, we know that medium-to-high priced epoxies are recognized for their quality adhesion, corrosion inhibition, and resistance to alkalis and solvents—yet our chemists are adamant that real world experience demonstrates that chemical versatility is not synonymous with corrosion prevention.
Adhesion Performance
Permeability Performance
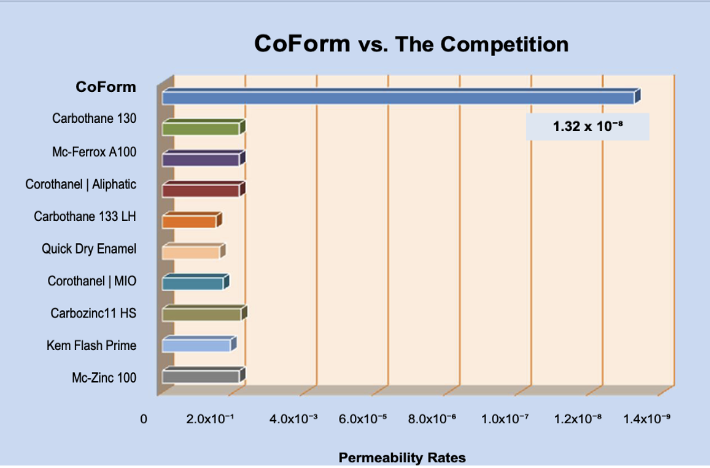
In fact, these coatings allow oxygen and moisture access to the metal substrate thereby exposing their true protection failures and mechanical limitations. They merely inhibit or postpone the penetration of oxygen and water or, like zinc-rich primers, they function sacrificially to delay not prevent corrosion; unless the zinc particles are housed in a binder, like CoForm, that is itself impermeable to oxygen and moisture. CoForm’s difference is not a coincidence, rather, an engineered difference that clearly eliminates all competition when corrosion prevention, control, and management are the mandated objectives.
How CoForm™ Differs from the Competition
Unlike competing coatings, CoForm creates and maintains an intimate bond with metal at its molecular surface level. CoForm was not
formulated merely to chemically bond to the surface of metal, rather to macro-encapsulate the surface thus forming an outer barrier layer.
The results of this chemical process are superior to those of typical “bonding” by other organic coatings. Tried, tested, and proven, CoForm
also can be sprayed directly over oxidized surfaces to contain any amount of corrosion on the metal substrate’s surface by simply depriving
the oxidation of oxygen, water, and moisture vapor.
Problems with Existing High-Performance Coatings
- Organic coatings are poor protectors of metal;
- Matching suitable coatings to unsuitable metal surfaces causes costly mismatches;
- Many factory-applied coatings cannot be reapplied, or repaired, once the metal is in service;
- Organic coatings fail to block elements that cause corrosion (i.e., oxygen and moisture);
- Sheer coating failures occur due to aging caused by UV exposure and weathering;
- Today’s coatings function only as temporary solutions to long-term problems.
- Superior Permeability Performance…. With no Global Competition
. . . additional problems include inferior bonding experiences
Significant amounts of chemical bond breaking within the polymer matrix weaken these coatings, which decreases their barrier properties and allows for the penetration of water vapor, oxygen, and ions. This results in disbonding or deterioration of coating-to-metal adhesion at the interface, which leads to corrosion and delamination.

Contact Info
95 Brown Road, Suite 270, M/S 1043, Ithaca, NY 14850
Phone: 607-266-0600
Email: marketing@precise-coatings.com